Most of the time, I aim to dial down parenting anxiety. Try not to think about it, or some version of that, is a commonly used refrain.
Today’s topic is almost the opposite. We’re talking about cytomegalovirus, or CMV, a virus that you may not have heard of, and that is a concern to pregnant people. The goal isn’t to scare you, though; it’s to inform and, ideally, empower. CMV is a complicated case where there isn’t a perfect solution, but there are ways to lower risks and ways to inspire policies that could improve outcomes. Don’t panic. Digest, inform, and react as appropriate.
For a more personal insight into CMV, a few years ago, I spoke with Megan Nix about her book Remedies for Sorrow, where she wrote about her journey with her daughter, Anna, who was born with congenital CMV. I would highly recommend the book, which is both informative and deeply affecting.

What is CMV?
Cytomegalovirus, or CMV, is a common herpesvirus. In adults and healthy children, the virus is typically mild or asymptomatic. Like most herpesviruses, the pattern of infection is an initial “primary” infection and then subsequent latent infection, which can cause flare-ups. (Think about genital herpes or cold sores.)
In parts of the developing world, virtually 100% of adults have been infected with CMV; in developed countries, it may be more like 50%. Still, it is extremely common.
Because CMV doesn’t cause manifest disease in most adults and children, you may not have heard of it. However, CMV is among the most prevalent causes of birth defects in the U.S.; CMV-related birth defects impact an estimated 1 in 1,000 births. These birth defects range from mild to severe or fatal, with hearing loss (in one or both ears) being the most likely. But even with this risk, pregnant people often do not hear about it until it may be too late.
CMV in pregnancy
The landscape of CMV in pregnancy is, frankly, confusing. The seriousness of the disease for the infant depends on the timing of maternal infection and transmission to the fetus. To help understand, I built a little visual, which I’m going to walk through in steps.
To begin: Infections with herpesvirus are classified in two ways. Primary infection is the first time someone is infected; non-primary are later flare-ups. During pregnancy, CMV infection in the mother can be either primary — this is their first exposure — or non-primary. In the U.S., an estimated 50% to 80% of prime-age adults have evidence of previous CMV infection. People in this group who got pregnant would have non-primary infection. For pregnant people without previous exposure, the yearly risk of developing a primary infection is estimated at around 2.3%. For pregnant people, young children in a household are a significant exposure risk.
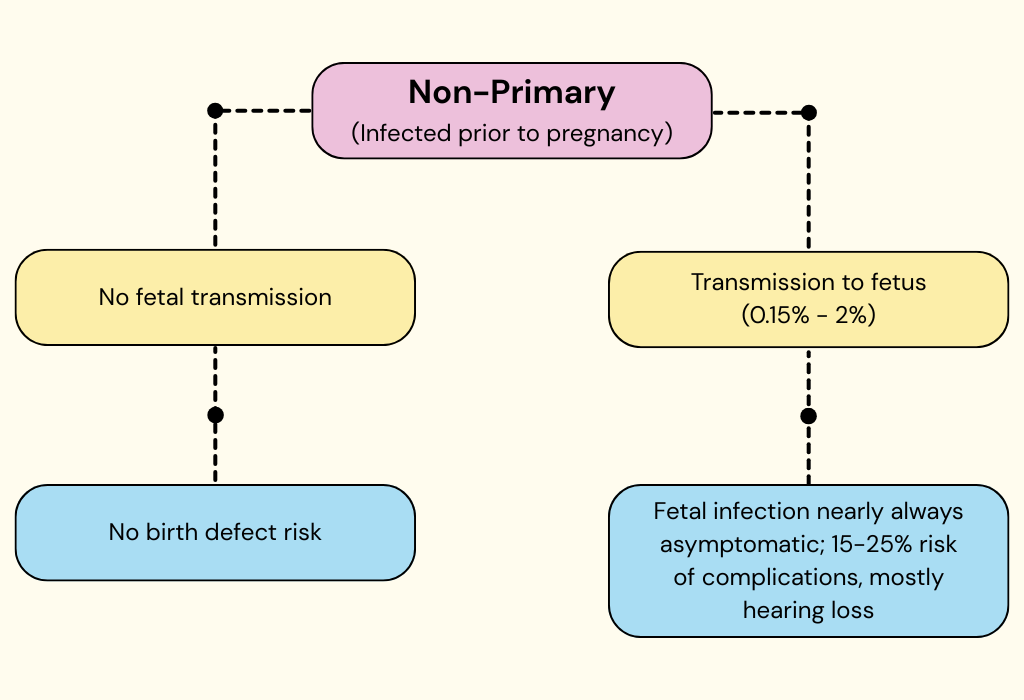
What this means is that most CMV infections in pregnancy are non-primary. The situation for pregnant people with a non-primary CMV infection is illustrated below. In most cases — 98% to 99% — non-primary infections are not transmitted to the fetus. In this case, there is no CMV-related birth defect risk. In an estimated 0.15% to 2% of cases, there is transmission. Nearly all fetuses are asymptomatic in these cases and generally do not show complications at birth. Unfortunately, an estimated 15% to 25% of these asymptomatic infections lead to later complications, most commonly hearing loss.
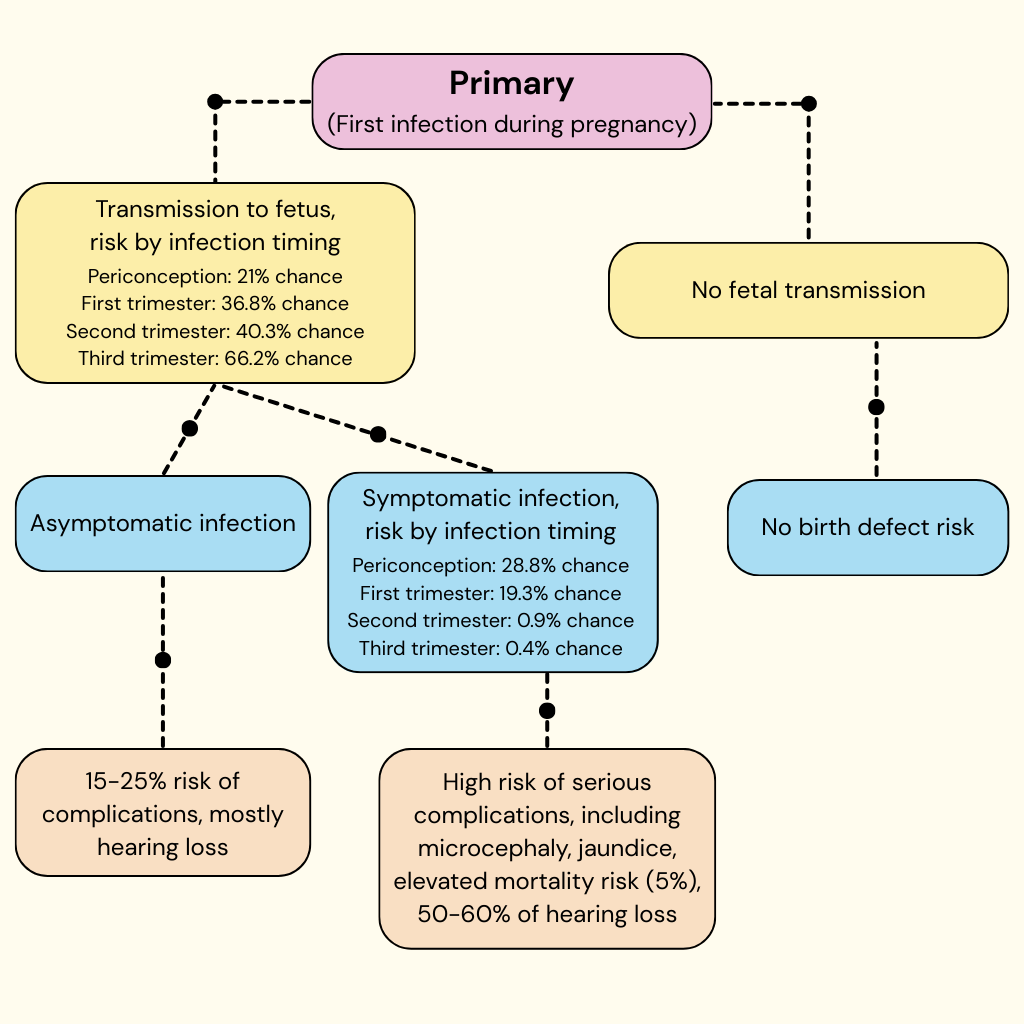
Primary CMV infection (infection among people who haven’t had it before) during pregnancy is much less common but is more serious, as illustrated in the (more complicated) portion of the chart below. As with non-primary infection, if there is no transmission to the fetus, there is no CMV-related birth defect risk. However, transmission to the fetus during primary infection is much more common and depends on the timing of infection during pregnancy.
Infection in the periconception period (four weeks before to six weeks after the last menstrual period) is estimated to result in fetal transmission 21% of the time, for example. Transmission risk to the fetus is highest in the third trimester.
Transmission to the fetus may then lead to asymptomatic infection; this would carry the same risks that it does when resulting from non-primary infection or symptomatic infection, which is much more serious. Symptomatic infection most commonly results from infection early in pregnancy or right before.
Putting the transmission and symptomatic numbers together, primary CMV infection is most dangerous in the first trimester, when approximately 7% of CMV infections would lead to symptomatic illness in the fetus. Infection in the third trimester leads to symptomatic infection only about 0.2% of the time. The complications from symptomatic CMV infection in a fetus are variable and can be very serious. The mortality rate is estimated at 5%, and the risk of hearing loss is more than 50%.
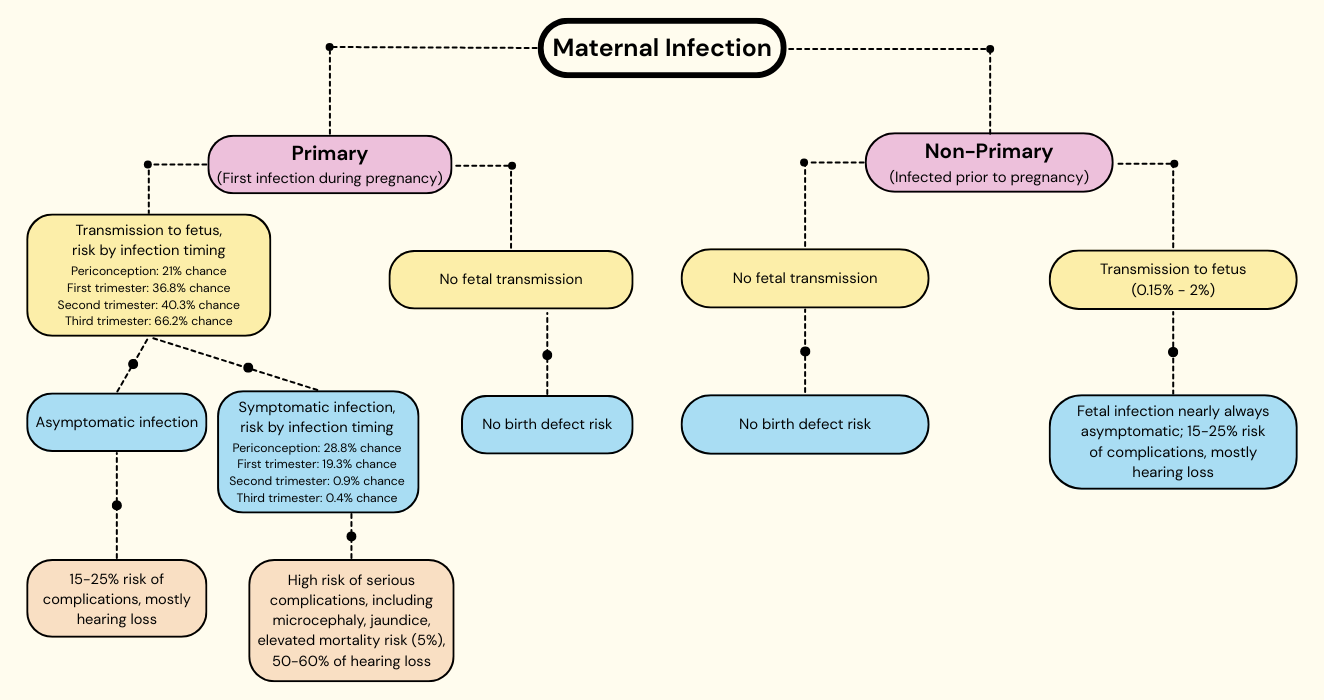
I put this all together in a single chart to navigate not so much what to do (more on that below) but how to think about it.
Can you lower CMV risk?
Given the serious and scary risks here, a natural question is: can they be lowered, and how?
The first best approach would be a vaccine, since CMV is a virus. Unfortunately, efforts to develop such a vaccine have not yet shown promise.
For now, we should look at other possible approaches to lowering risk. The first thing to recognize is that the risk of CMV infection is much greater if you have never been infected before (that is, a primary infection). The degree to which you want to undertake mitigation, then, should depend on this status.
How would you know if you were in this group? Testing. There are simple laboratory blood tests for CMV infection. They are not routinely ordered for pregnant people, which means both that there may be reluctance to order this testing and that insurance may not cover it. But you can ask.
There are good reasons why doctors may be reluctant to screen, largely reflecting the fact that there is not much to do, and either a positive or negative test can be scary. So this isn’t an obvious choice in either direction; it may depend on your own preferences.
If you haven’t been infected in the past, or you don’t know, there is the approach of avoidance. For parents, small children are the most common vector of CMV; primary infection occurs about 25% of the time with a child shedding CMV, versus only 2% if they are not. Children can shed the virus for a long period of time after infection, and without a test (possible, but very unlikely to be something you can access), it is hard to know if it is happening.
The main thing you can do for avoidance is enhanced hygiene behaviors. This includes hand washing and avoiding contact with saliva. There is some limited evidence in favor of these, and the logic is sound, given how we know the virus spreads. I hear what you are telling me: avoiding your sick toddler is impossible. That’s true! But there are small, simple changes you could make. For example, do not kiss them on the mouth. Do not eat food they have bitten. Do not share their straw. When you help them blow their nose, wash your hands after.
None of these is going to be a foolproof way to avoid illness, but they can make a difference, and risk reduction is just that — reduction. And this reduction may well be worth it, especially if you have not had CMV before pregnancy.
What if you are exposed to CMV while pregnant?
Primary CMV infection during pregnancy (or other times) appears, typically, as a flu-like illness. If you have such an illness, and if flu and COVID tests are negative, a CMV test is important. Pregnant people who have primary CMV infection should be carefully monitored for the possibility of complications with their infant.
If this occurs, your doctor will be your best asset. An amniocentesis is common to test for fetal transmission, and frequent ultrasounds can detect physical abnormalities. Your baby will be more closely monitored both before and after birth.
There is an antiviral treatment that has been shown in randomized trials to improve outcomes for impacted infants if it is used within the first month of life (the drug is ganciclovir in IV form, or in oral form, valganciclovir).
This antiviral is recommended for symptomatic CMV and for asymptomatic CMV with primary hearing loss. That is, an infant with CMV who has one or more symptoms (microcephaly, poor muscle tone, petechiae [tiny red spots], jaundice, and several others) and a positive CMV test, or an infant with hearing loss and a positive CMV test, should be treated.
Given this treatment, there is value in screening infants who have any of these symptoms early to see if CMV is a cause. In the podcast episode I linked above, Megan and I talked about policies that would make this part of the standard newborn screen. This is a policy that would need to be introduced at a hospital or state level, as it has been in Minnesota.
Other states have introduced targeted newborn screening based on symptoms. Even if your state does not mandate this, as a parent, it is something to be aware of and something you could ask for, especially if your child shows any CMV symptoms at birth. This document, created by Dr. Ted Maynard and the Colorado Chapter of the American Academy of Pediatrics, may be helpful in guiding a conversation with your provider.
If you’d like to know more, head to the National CMV Foundation. If your pregnancy, baby, or child has been affected by CMV, there are support groups that can be accessed through it, too.
The bottom line
- CMV is an extremely common, usually mild virus in adults and children, but it is mainly a concern during pregnancy because it is a leading cause of serious birth defects.
- The risk of CMV infection is much greater if you have never been infected before, so requesting a test to learn your status is a good idea.
- If you haven’t had CMV before or you don’t know your status, the main thing you can do to minimize your risk is enhanced hygiene behaviors (hand washing, avoiding contact with saliva). At this point, there is no vaccine for CMV.
- If you are pregnant and have flu-like symptoms, and flu and COVID tests are negative, you should be tested for CMV. If the test is positive, your baby will be more closely monitored both before and after birth. If your baby is affected, early antiviral treatment can significantly improve outcomes.




















Log in
The prevention measures listed here for parents of young children miss a really big one: wash hands after contact with urine / changing diapers. Urine is an especially concentrated source of CMV. It is likely how I acquired my primary CMV infection when I was in my first trimester with my second pregnancy last summer. I had read this page, gotten my titers done and knew I was seronegative for CMV, and was taking a ton of hygiene precautions around my daughter’s saliva and boogers, but I wasn’t thinking about pee diapers. A pediatric virologist I spoke to later went so far as to recommend that I wear gloves to change my daughter’s diapers in a future pregnancy. Please update this article so that people know about the urine risk!
It says “Transmission risk to the fetus is highest in the third trimester.” But the information following is contradictory. Should this say it’s lowest in the third trimester?
Nvm! I realized I read that wrong. Transmission is highest in 3rd but infection most serious if it happened in the preconception or first trimester period.